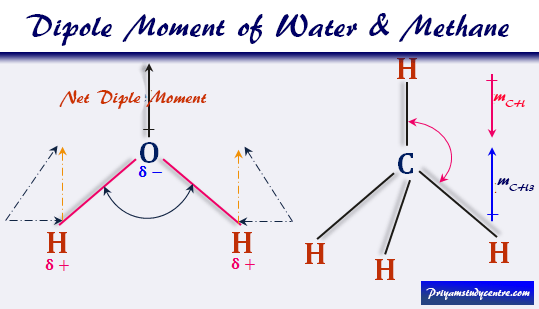
Molecules with zero dipole moment are non-polar, while molecules with dipole moment are said to be polar. In finding the polar nature of the bond: As the magnitude of dipole moment increases, more will be the polar nature of the bond. In the diatomic molecule of HCl, the dipole moment of the HCl molecule is the same as dipole moment of HCl bond, which is 1.03D. However if the distance between the proton and electron increases and they get far apart from each other, the dipole moment increases. When a simple system is considered where a single electron and a proton is separated by a fixed distance, if the distance between the electron and the proton is smaller and they are close together, the dipole moment of the degree of polarity decreases. The orientation of the dipole is along the axis of the bond. The bond dipole is interrupted when the charges separated over a distance r are between the partial charges Q+ and Q− (or the more commonly used terms δ+ - δ−). When each of the bond electronegativities are added together, the dipole moment points in the direction of the vector quantity of each of the bonds. So as to establish a polar bond, the difference in electronegativity needs to be large. Therefore the larger the difference between the electronegativity of the two atoms, the more will be the electronegative activity of the bond. The vector always points from positive to negative in the individual bond dipoles and also on both the molecular dipole moment. Thus, water molecules are considered polar in nature. Fo r example water (H 2 O) as a lone pair of electrons on the oxygen atom and its structure according to the VSEPR theory is bent and thus the vectors representing the dipole moment of each of the bonds do not cancel each other out. 
The dipole moment acts in the direction of the vector quantity. R i is the vector representing the position of i th charge. Q i is the magnitude of the i th charge, and The dipole moment of a molecule can be calculated by another primary Equation that is mentioned below: It is measured in Debye units, represented by D.ĭ = 3.33564 ×10-30 Cm C = Coulomb, m = meter. Where, μ is the bond dipole moment, Q is the magnitude of the partial charges □+ and □–, and d is the distance between □+ and □–. It is given by the equation:ĭipole moment (µ) = Charge (Q) × Distance of separation (d) Dipole moment definition can be given as the product of magnitude of electric charge of the molecule and the internuclear distance between the atoms in a molecule. The dipole moment ( μ ) is also used to determine the size of the dipole.dipole moment which is equal to the distance between the charge X the charge is measured in the Debye unit where 1 Debye = 3.34×10 -30 Cm. The dipole moment is established when the two electrical charges that are of equal magnitude but of opposite signs are separated by a distance. The arrow signifies the shifting of electron density in the molecule. Bond dipole moment is considered as a vector quantity, as it has both magnitude and direction. This measurement of polar character of a chemical bond in a molecule, between two atoms, is given by bond dipole moment. Polar character is the separation of positive and negative charges, in a compound. Thus the difference in the electronegativity combined with the presence of the lone pair of electrons on the oxygen atom gives it a partial negative charge and the hydrogen atom the positive charges. One of the most common examples is the water molecule that consists of one highly electronegative oxygen atom and two electropositive hydrogen atoms. It also happens with the atom bearing the lone pair of electrons and the difference in the vector points of the electronegativity in a similar way. This usually occurs when one atom is more electronegative than the other atom which results in more pulling of the electron cloud by the higher electronegative atom. Therefore in other words, the dipole moment is created when the atoms of a molecule share the electrons unequally. Dipole moment is actually the measurement of the polarity of the molecules. In the size of the dipole moment the distance of the bond also plays a crucial role in determining the magnitude of the dipole moment. 
The main reason for the rise of the dipole moment is due to the difference in the electronegativity of the atoms of compounds formed. The main cause for the development of the dipole moment is the electronegativity difference between chemically bonded atoms or elements.ĭipole Moments mostly occur between two ions in an ionic bond or between two molecules when they share a covalent bond. Both ionic and covalently bonded compounds develop dipole moments. The separation of charges in any system leads to a dipole moment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
